Structure and dynamics of biomacromolecules dictate their functions. Our group is interested in understanding the molecular mechanisms of protein complexes through detailed characterization of their structures and dynamics. In particular, we are fascinated by supramolecular assemblies/molecular machines which play a central role in maintaining proteostasis in the cell, such as the proteasome and various AAA+ unfoldases and proteases. We use advanced Nuclear Magnetic Resonance (NMR) techniques to study the structural and dynamic details of proteins, and explore the correlations between structure/dynamics and function. We seek to uncover how these complex molecular assemblies work, shed light on how certain mutations in these proteins cause diseases, and provide strategies on intervening their functions by therapeutics. We apply various biophysical and biochemical methods, as well as other powerful structural tools such as cryo-EM and Mass Spectrometry through collaboration, to achieve our goals.
Mitochondrial AAA proteases
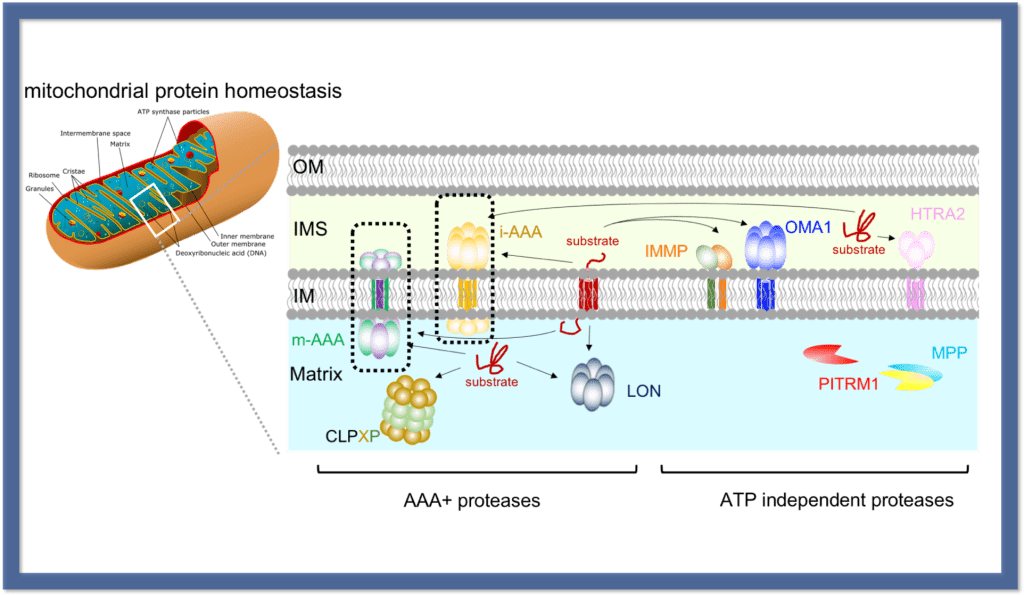
Mitochondria is the ”power plant” of eukaryotic cells, and plays prominent roles in a number of metabolic and signalling pathways. Functions of mitochondria rely on proteostasis in the organelle which is maintained via an intricate network of mitochondrial protein degradation machines (see figure). Among these proteases, there are two AAA proteases (i-AAA and m-AAA, in dashed boxes) that play essential roles in mitochondrial homeostasis. They provide surveillance on both surfaces of the mitochondrial inner membrane by removing damaged/misfolded proteins. They also regulate mitochondrial biosynthesis, metabolism and dynamics by specific proteolysis. Our work seek to gain structural and dynamic insights on these two proteases and unravel mechanistic details about how they function.
AAA+ ATPases
AAA+ ATPases are a versatile class of molecular machines which transform chemical energy of ATP hydrolysis to mechanical forces in order to remodel substrates and achieve various cellular functions. We are currently interested in exploring the category of AAA+ proteins that are involved in unfolding protein substrates and preparing them for proteolysis, such as VAT (see publication 12,13) and p97 (see publication 19). We would like to understand the interplay between these molecular motors and their substrates as well as their cofactors using NMR and various other techniques.